Steroids
CTS is clearly a steroid responsive condition. Randomised controlled trials have repeatedly shown short term benefit from injection of steroids into the area of the carpal tunnel (Dammers 1999) and improvement has also been shown after a short course of oral steroids (Chang 1998). Such trials are almost redundant however (the ‘parachute’ argument) because the short term effect of steroid injection is so dramatic with approximately 80% of patients being markedly improved within days of a single injection at the wrist. The things we really need to know about steroid injection for CTS are:
- How does it work (We don't know but read the section on a theory of CTS for a possibility?
- How long does the effect last? (see notes on Visser 2011 below)
- How risky is it?
- Who is most likely to respond well to treatment?
- What is the most effective dose and approach with the needle, and is it worth using ultrasound guidance?
- Is it worth repeating them, and if so how many times?
- Does injecting first prejudice the outcome of later surgery?
... and in answering these questions the existing scientific literature is surprisingly uninformative. The original thinking behind trying steroids for CTS seems to have been based on the theory that CTS was due to inflammation of the flexor tendon sheaths and that a powerful anti-inflammatory (which steroids are) would reduce this, but as we have seen in discussing causes, most cases of CTS do not have inflammation of any kind in the carpal tunnel. Only one good trial of steroids (Atroshi 2013) has satisfactory follow up extending beyond 3 months and even unsatisfactory follow-up arrangements in published trials do not extend beyond 2 years at most. I have met two patients however who seem to have had a remission after one steroid injection lasting 48 years. No conceivable follow up study would have included these two patients as late relapses and the question of whether injection is ‘always’ a temporary fix for CTS if you wait long enough is going to remain unanswered. In practice of course the distinction between a ‘temporary’ cure lasting 48 years and a ‘permanent’ cure is an academic one and, as with splinting, if CTS is in some cases a temporary problem anyway, then a temporary solution may be appropriate. At a more practical level, our own data in Canterbury suggest that approximately half of the patients initially treated with a combination of splints and steroid injection will return for further treatment within the first year after injection. Beyond this point it is clear that patients continue to relapse after injection whereas patients treated surgically who are fully recovered one year after surgery rarely relapse.
A recent observational study has provided some figures for relapse after a first injection which appear to be comparable to our findings in Canterbury (Visser 2011). This group was able to follow up 211 of 273 patients who were initially treated with injection of 40mg methyprednisolone so their follow-up rate of 77% is quite good for a study of this type. The patients who were still satisfied with the outcome were as follows:
6 months - 132 (63%)
12 months - 102 (48%)
18 months - 71 (34%)
They found that the length of remission depended to some extent on the neurophysiological severity of the CTS before injection, as follows:
'mild' (Canterbury grades 1 and 2) - median time until relapse 15 months (60 patients originally injected)
'moderate' (Canterbury grade 3) - median time until relapse 5 months (203 patients originally injected)
'severe' (Canterbury grades 4-6) - median time until relapse 4.5 months (10 patients originally injected)
Gender, side and clinical severity of CTS were not predictive of the length of remission. They do not comment in the text on patients who did not respond to the initial injection at all but a figure in the paper suggests that a little over 30% of all the patients were considered relapsed within the first 6 weeks or so of injection, comparable with other studies. They conclude by suggesting that about 50% of patients with mild neurophysiological abnormalities can expect to remain in remission for 15 months or more.
A variety of approaches can be made with the needle with the aim of placing steroid safely somewhere close to the carpal tunnel.
With the advent of high resolution ultrasound imaging of the carpal tunnel there has been considerable interest in performing injections under real-time ultrasound guidance. Given the apparently very low risks of carrying out injection without such technical assistance it is not clear that this is really necessary. However findings like this on ultrasound...
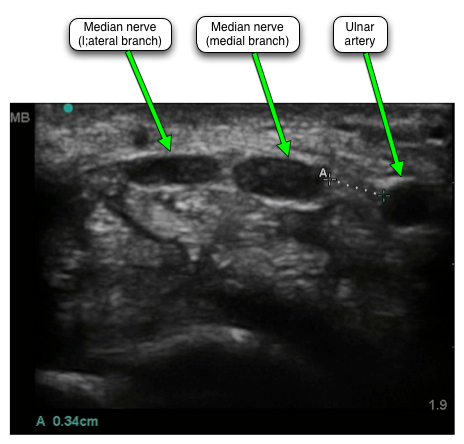
... where the gap between the ulnar artery and the nearest of the median nerve branches (marked A) is only 3.4 mm and where the median nerve itself has two branches spread across a horizontal distance of about a further centimetre, provide strong encouragement to take a look inside the wrist to spy out the anatomy before inserting a needle. The often very close proximity of the ulnar neurovascular bundle to the median nerve was actually highlighted in one of the earlier ultrasound studies (Nakamichi 1998) where the gap was found to vary from 2.8 to 11.2 mm.
A small study of 21 patients published so far only as an abstract (Kirschner 2011) claimed that patients injected under ultrasound guidance had greater pain relief at 2 and 8 weeks after injection than those injected by a traditional 'blind' method, the implication being that more precise placement of the steroid was more effective, not that the ultrasound guided method was any safer. This study was described as 'single-blind' but it is hard to see how either the doctors or the patients could be unaware of difference between ultrasound guided and traditional injections so this result appears rather doubtful.
In other studies of injection the short term success rates are all very similar regardless of the exact method and site of injection. One small study directly compared a traditional injection site just ulnar to the tendon of flexor carpi ulnaris, 3-4cm proximal to the wrist crease (12mg methyprednisolone + lidocaine) with an injection given 2-3cm distal to the middle of the wrist crease (35mg methyprednisolone + lidocaine). Success rates at 1,3,6 and 12 weeks after injection were not significantly different between the two groups - 57% at 12 weeks (Habib 2004).
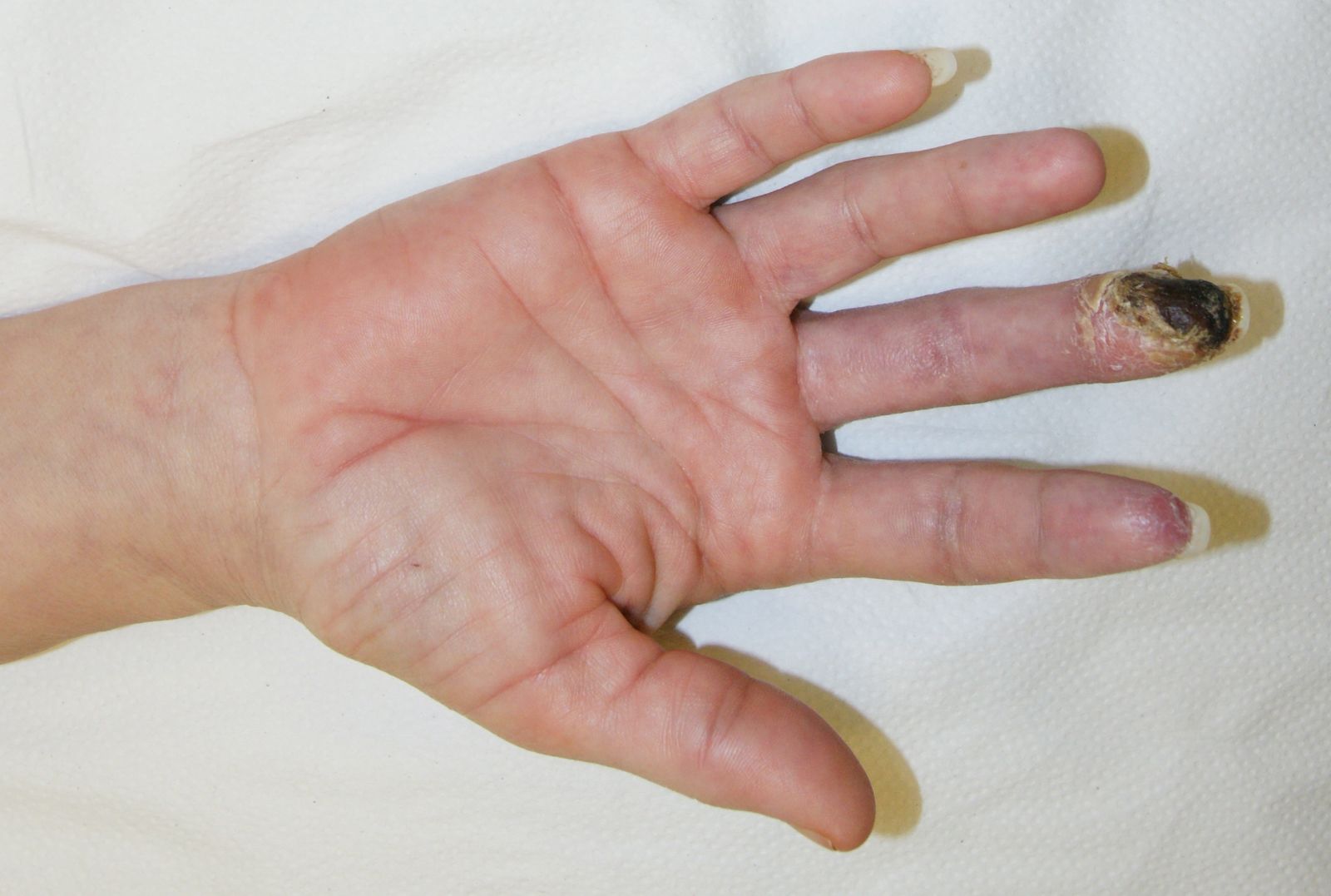
- much is made in the surgical literature and in casual conversations with surgeons of the risks of steroid injection at the wrist and almost every surgeon has one ‘horror story’ of a patient seriously injured by this procedure who subsequently had to be rescued (with varying success) by the surgeon. In fact well documented instances of such injuries are surprisingly rare. In the published literature there are instances of injury to the median nerve itself from direct (intraneural) injection and I have been able to find 12 of these in total worldwide. I have seen this myself once into the median nerve and once into the ulnar nerve, and we have also seen two examples of vascular complications resulting in gangrene of a finger tip.
There are now two other case reports of this complication (Hussain 2011, Payne 2008). In one of these the injection was triamcinolone with lidocaine and in the other the exact components of the injection were not stated. Payne et al believed that the digital ischaemia which they witnessed might have been due to micro-embolisation of the small arterioles in the fingers by particulate matter contained in the steroid injection but Hussain did not suggest a mechanism for their case.
There are anecdotal examples of injuries to the flexor tendons and I know of one report of flexor tendon rupture after many injections but the details of this case are a little vague. We have seen one definite example of rupture of a long flexor tendon to the little finger after ten injections to that wrist and also a possible flexor tendon rupture occurring several weeks after a single carpal tunnel injection, but tendons do sometimes rupture spontaneously and this second case may not have been related to the injection. Presumably many such complications go unreported but even so the lack of well documented cases is striking. Possibly the best way to assess the risk is to look at the published trials of steroid injection, in all of which there has been systematic reporting of side effects. When pooled, these trials have involved well over 3000 injections and in none of them have there been ANY serious injuries related to the injection. We have now done enough injections in East Kent (over 5500) to have a fair idea of the rate of significant complications, of all kinds, and this seems to be in the region of 1 per thousand injections, clearly much less than the surgical complication rate. Local steroid injections for CTS do not cause systemic steroid side effects (see the section on oral steroids below)
Blanching/depigmentation
With superficial steroid injections loss of pigment in the skin at the site is well recognised but this is relatively rare after carpal tunnel injection where the injection is placed fairly deep in the tissues. This is not to say however that it never occurs. It may take up to 2 months to appear and is presumably due to the steroid tracking back along the needle path to the skin.

The loss of colour is more obvious here of course because of the underlying skin colour. The normal colour may return over weeks or months and 'most' patients have recovered by 12 months. Exposure to ultraviolet light may help.
Bruising - a few patients report bruising around the injection site after injection, which can take a week or more to clear afterwards.
Facial flushing - we have met thirteen patients (12 of them female) who reproducibly experience marked facial flushing for a day or two after steroid injection.
Menstrual irregularities - a few patients have reported short term disturbance to their menstrual cycle after injection.
Pain on injection - Local steroid injections are widely used in rheumatology and orthopaedics for a variety of joint and soft tissue disorders. Some of these injections have a reputation for being painful. The frequency of severe pain on injection seems to vary somewhat depending on the site of injection and injections for CTS are generally not very painful. The key word here however is ‘generally’ - occasional CTS injections are VERY painful and this seems to be entirely unpredictable - so much so that patients who have had both wrists injected at the same time, by the same doctor, with the same steroid will sometimes report that one side was painful while the other was not. 90% of injections are not very traumatic for the patient though there is usually some local discomfort for a few hours afterwards. My own patients tend to report the pain from local steroid injection as 1-3 on a pain scale from 0 - no pain at all, to 10 - the worst possible pain. If you would like to report your own experience with injection you will eventually be able to do so through registering on this site. It is possible that eventually I may be able to work out why some injections are painful and some not, given enough data.
A slightly different issue to the pain of the injection itself is the possibility that, when injections are used for soft tissue inflammation, there may be a few days of local swelling and increased pain after injection. This reaction has been called 'steroid flare' but it is not clear whether it results from physical/chemical irritation or some other mechanism, nor is it clear whether it is anything to do with the steroid itself or is related to other components of the injection. One paper exploring the chemical irritation aspect looked at whether the acidity of the injection had anything to do with it but found no difference in the incidence of the flare reaction between acidic and neutral injections (Goldfarb 2007). The injections used in this study contained two local anaesthetics as well as the steroid so once again the exact chemical component causing the reaction remains obscure and the injections were being given for DeQuervain's tenosynovitis or trigger digits, not carpal tunnel syndrome. They did find quite a high incidence of 'steroid flare' though, defined as a 2 point increase in pain on a 0-10 visual analog scale compared to the pre-injection pain level at any time during the 7 days following injection. 41 of the 125 patients met this criterion for flare. This response to steroid injection seems to be relatively rare when injections are used for carpal tunnel syndrome and is always transient, settling in a few days.
A more recent study of patients having injections for trigger digits (Julka 2012) tried to study factors which might predict pain both at the time of injection itself, and during the next 24 hours (looking for the phenomenon of 'steroid flare'). They used the familar 0-10 subjective pain scale where 0=no pain and 10=the worst pain imaginable. For trigger digit injections the mean pain score was 4.3 with a range from 0 to 10! and the average pain score 24 hours later was 1.8 with 17/100 patients reporting pain the following day which was equal to or greater than the pain experienced with the injection itself - a possible indicator of 'steroid flare'. The best predictor of pain on injection was the patient's pre-injection expectation - ie those patients who expected it to hurt a lot tended to experience more pain. Females and patients with high scores on a questionnaire designed to detect depression also had higher pain scores. The only predictor of next day pain was the pain experienced at the time of the injection. It may well be, therefore, that patients who do experience a lot of pain on injection might be causing others to experience more pain on injection by going around telling everyone what a terrible experience injection was!
Which steroid and what dose? - Steroids are in one sense ‘natural’ in that the body produces a variety of steroid hormones itself. The word steroid itself refers to a common chemical structure which is shared between a variety of different hormones (4 carbon rings, three of 6 atoms and one of 5 atoms) As well as the naturally occurring ones there are a range of synthetic steroids which share the same common chemical structure and which have similar biological effects to the naturally occurring steroid hormones. A variety of different steroids have been tried in CTS, all of which are analogues of the naturally occurring chemical hydrocortisone, differing mainly in terms of potency (the biological effect per milligram) and physical properties like solubility. So far as clinical effect on CTS is concerned there are only a few rather unsatisfactory trials comparing different doses and steroids. (O’Gradaigh 2000) showed no significant differences between 25 mg or 100mg of hydrocortisone and 20 mg of triamcinolone. The Dutch group who carried out one of the early high quality trials of injection have subsequently looked at some variations on steroid injection and found no significant differences at one year between 20, 40 or 60 mg of methylprednisolone but they did suggest, from supplementary analysis of patients in their other studies, that patients given a second injection might do better overall(Dammers 2001, Dammers 2006). (Wong 2005) found no difference between one injection of 15mg methylprednisolone and two such injections 8 weeks apart. There is thus no particular reason to choose one steroid over another, nor is there any overall agreement about what is the best dose. Some steroid preparations for injection use are designed to stay in the same place for some time after injection but whether these are more or less effective in CTS is unknown.
A study comparing 40 and 80 mg of methylprednisolone has just been completed in Sweden (Atroshi 2013) and is probably one of the most important steroid trials to date. It is not perfect and its results are open to some different interpretations but the bottom line is that about three quarters of a group of patients initially treated with a steroid injection had had surgery within one year compared to 9 out 10 patients in the placebo group. My thoughts on these results are set out in full in the page of this site dealing with important trial evidence. The results do not suggest that there is a dramatic benefit in using a higher dose, at least not in an initial injection.
With or without local anaesthetic? - it has been the practice of some doctors to include in the same injection with the steroid preparation a local anaesthetic agent too - usually lignocaine/lidocaine. This does not seem to appreciably reduce the already usually modest pain associated with injection and has no therapeutic purpose. All it does is leave the median nerve innervated part of the hand anaesthetised until it wears off, rather like a dental anaesthetic. it has been argued that that this numbness in median nerve territory confirms that the injection has been put in the 'right' place in the carpal tunnel but as it does not seem to matter in terms of therapeutic effect exactly where the injection is placed this seems to be largely irrelevant. We do not add local anaesthetic.
Oral steroids - One trial has shown that CTS responds to a short course of oral steroids (Chang 1998). This clearly avoids any risk of injection injury but oral steroids have a deservedly bad reputation for side effects. Regular large doses of steroids administered to the whole body cause weight gain, diabetes, osteoporosis and thinning of the skin and are highly undesirable long-term treatments. Given that a single local injection, entailing a tiny overall systemic dose of steroids insufficient to cause any of this, is so effective in CTS there seems to be no good reason to use oral steroids.
Treatment with more than one injection
When we start to consider second injections we largely leave the realm of evidence based medicine. There are a reasonable number of good quality controlled trials looking at the short term effectiveness of the first injection for CTS. Until recently there were NO trials of second and subsequent injections though rheumatologists have been giving second and third injections for CTS for years. In his first large summary paper on CTS (Phalen 1966) George Phalen included a table summarising the experience of 270 wrists treated by injection with the number of injections per wrist ranging from 1 to 11. Only 60 (22%) of these hands had eventually come to surgery and the only mention made of serious complications is mention of a patient given 17 injections in the left wrist and 11 on the right who developed 'chemical neuritis' which seems to have improved, but not resolved, spontaneously. The text says that if there is no response to the first injection there is unlikely to be any benefit in giving a second but the numbers underlying this statement are not given. After this paper there is a long silence on the subject of multiple injections though George Phalen did update his data table to include 497 wrists in a subsequent publication (Phalen 1972). There are anecdotal reports of patients who have been maintained for long periods with an occasional repeat injection, usually at intervals of 6 months to two years, without there being any overall deterioration in their CTS. There are also anecdotal reports suggesting that very large numbers of injections into the same carpal tunnel might have deleterious long term consequences.
There are four trials directly comparing injection with surgery, three of which concluded that surgery was better (Demirci 2002, Hui 2002, Ismatullah 2013), the other of which found they were equally effective at one year after treatment (Ly-Pen 2005), but that relapses continued to occur in the injection group after the one year initial follow-up (Ly-Pen 2012). Taken together these studies suggest that surgery has a greater impact on symptoms overall than a single injection and that the effect is much more likely to be permanent with surgery. However these studies rather fail to address the fact that a small but significant number of patients get very bad results from surgery whereas major complications of injection are extremely rare and none of them are reported in a way which allows one to say how many patients might have been spared surgery by the use of injection.
Perhaps encouraged by the results of the existing trials there is now some evidence that people are starting to take more interest in the possibility of using steroid injection as a viable long term strategy for managing CTS. A Scottish group has looked at the 5-year outcomes of 824 patients with CTS who initially opted for steroid injection as their first treatment rather than surgery (Jenkins 2012). 15% of these patients had required carpal tunnel surgery by one year and 33% by 5 years, suggesting that the majority of these patients had NOT gone on to require surgery over quite a long period. Female patients, those with diabetes, and those with abnormal nerve conduction studies at diagnosis were found to be more likely to require surgery in the long run. The approach to the neurophysiological studies however was relatively unsophisticated, merely characterising them as normal or abnormal and 50 patients were not tested at all. 90% of the patients who were tested had abnormal results, though the neurophysiological criteria for abnormality are not described in detail in the paper.
We have recently repeated a version of this study in Canterbury (Hameso 2017). We made more determined efforts to find out exactly what had happened to all 254 patients who started treatment with steroids in 2007 during 2015, 8 years later. We eventually concluded that only 41% of the patients had required surgery during that 8 year period. The un-operated patients had had between 1 and 10 injections, with an average of two.
Further supporting this picture on long-term outcome, a study from the Mayo Clinic (Evers 2017) found that 32% of patients given an initial steroid injection had not required any further intervention during a median period of follow-up of 7.2 years. This study also found that outcomes were slightly worse with more severe neurophysiological abnormalities. They looked for other predictors of outcome but the only statistically significant findings were that patients with rheumatoid arthritis did better, and that the use of a larger volume of injectate (even containing the same dose of steroid) was associated witha better outcome - a curious finding that requires further investigation. 131 patients in this study had a second injection but there is no mention of any third injections.
Another study with rather different methodology looked at 120 patients treated with one, two or three steroid injections over the course of 1 year (Berger 2013). The patients had electrodiagnostically confirmed CTS but no exclusions seem to have been made on grounds of severity of CTS and 36 of these patients had absent median sensory nerve action potentials which would suggest that a fair number of them had quite severe CTS (probably at least grade 4 on the Canterbury scale). A satisfactory outcome was defined as being ‘symptom free to the extent that no further treatment with injection or surgery was necessary’ and was achieved at 1 year by 30 patients with one injection, 11 patients with two and a further 5 patients after a third injection – thus 38% of these patients were successfully treated by injection alone 1 year after beginning treatment. What is unclear about this study is the way in which decisions were made about second and subsequent injections. So far as I can tell from reading the paper, all patients were offered the option of a second injection, with the decision being left largely to the patient. A third injection however seems only to have been offered if the second one had been very effective for at least a while.
In this study the quality of the initial response to injection had some predictive value for the eventual outcome. 54 subjects had a very good initial response to the first injection and this group did rather better at one year overall with 52% remaining in remission compared to the 38% overall figure.
It is also worth noting that, as with the Scottish study, the majority of these patients (62%) had NOT been referred for surgery after being followed up for 1 year, suggesting that they considered their symptoms adequately controlled.
Finally, we have just published a slightly different way of looking at this based on patients treated in Canterbury (Ashworth 2013). We identified a group of 229 patients who had had two injections into the same hand and looked to see whether the initial responses to second injections were any different to the initial response to the first, either in terms of quality of relief of symptoms or duration of effect - ie we were looking for evidence of steroids either losing effectiveness with repetition, or conversely, becoming more effective with cumulative dosing. We found neither. Second injections were, on average, just as effective as first injections. We found no reliable way of predicting the outcome of the second injection, even when we knew the outcome of the first one but the effects of the first and second injections were somewhat correlated.
All three of these studies probably suffer from the same problem. We all tend to give up on therapeutic approaches which are not obviously successful. Thus if a patient reports no response to a first injection, or only slight benefit, we tend not to try a second one. When, as in the above studies, decisions about further treatment are influenced by the initial injection response this introduces a severe bias into the outcomes. It has not however been conclusively shown that a second attempt at injection is predestined to fail just because the first one failed, and indeed we found a few patients in whom a second try was successful despite a poor response to the first injection. It may therefore be necessary to carry out a trial in which all patients are given a second injection when they have persistent or recurrent symptoms, even if there seems to have been no response to the first. Only then will we learn the true value of a second try.
Does injection make it less likely that surgery will work later on?
Some surgeons believe that patients have better outcomes if they have surgery immediately rather than trying injection first and then having surgery later. There is one, rather poor quality, retrospective patient survey which claims to support thsi view (Vahi 2014). We have just published what we think is a rather better study of the question in 942 East Kent patients, 664 whose first treatment was surgery and 278 who had one or more injections first but still ended up having surgery (Bland 2015). Surgical outcomes were assessed 3 months after the operations by overall patient satisfaction and by the SSS/FSS questionnaire as used on this website.
There were no significant differences in outcome. 85% of the direct to surgery group and 84% of the prior-injection group reported themselves either "much improved" or "completely cured" after surgery and there was no significant difference between the groups in post-operative SSS and FSS scores. 3.3% of the direct group and 3.9% of the prior-injection group felt they were worse off after surgery - again not a significant difference.
Overall we feel confident that injecting first does not, in itself, prejudice the outcome of subsequent surgery. However we would still suggest caution in following up patients after injection. If the symptoms have not been alleviated significantly by injection then further treatment should be sought. When injection does work well, patients should seek further advice quickly if and when symptoms recur.
There is a detailed FAQ list relating to the way we have chosen to implement injection treatment in East Kent.
Revision date - 15th August 2017